TK-NOG マウス
TK-NOGマウス
次世代重度免疫不全 TK-NOG マウス

| 系統名(カルタヘナ情報) | マウス TK-NOG Tg |
|---|---|
| 系統名(略称) | TK-NOG マウス |
- ご購入にあたりまして使用同意書へのご署名が必要となります。
- またこのマウスモデルのご使用はカルタヘナ法の研究開発に係る遺伝子組み換え生物等にあたります。
弊社より情報提供書をお送りいたしますので、ご確認のうえ各機関のルールに従ってご使用ください。 - TK-NOGマウスは、日本国内で生産しお届けします。
- TK-NOGマウスをご購入後、お客様の施設で臓器等の再生医療製品の移植実験等実施することができます。
開発
マウスAlbプロモーター下流に人工イントロン、HSVtk 遺伝子を配置したベクターを(NOD♀ x NOG♂)F1 マウス前核期受精卵へマイクロインジェクションし、得られたトランスジェニックマウスをNOGマウスへバッククロスして作製しました。
特徴
- TK-NOG マウスは NOG マウスにさらに遺伝子導入したマウスで、アルブミン遺伝子エンハンサー/プロモーター支配下にHSV-TK (Herpes Simplex Virus-1 Thymidine Kinase)が肝臓に発現し、ガンシクロビル投与によって肝臓特異的な障害を誘導するようにデザインされたマウスです。
- 肝傷害を発症したマウスにヒト正常肝臓細胞を脾臓門脈経由で移植すると、ヒト肝臓が再構築され、いわゆるHu-liver TK-NOG マウスが得られます。
研究用途
- Hu-liver TK-NOGマウスの作製 移植方法はこちら
- iPS由来肝臓組織の移植モデル等
生産と供給
- 日本クレア株式会社に生産を委託し、日本国内で生産しています。
- AAALAC認証を含む国内第三者認証をうけた生産施設で生産しています。
日本クレア株式会社動物福祉への取り組みはこちら - 動物の輸送は、温度管理された専用車両により行います。
ヒト化 Hu-liver TK-NOG マウス
特徴
- Hu-liver TK-NOG マウスではアルブミンの他、ヒトの補体 C3 やトランスフェリン、セルロプラスミン等、重要な血清タンパク質についても合成・分泌が行われていることが確認されています。またマイクロアレイによる網羅的遺伝子発現解析では遺伝子発現プロファイルが移植したドナーの肝細胞と再構築した肝臓で高い相関が認められ、ドナーの肝細胞の性質が再構築した肝臓に反映されることも確認されています。
- 薬物代謝酵素についても再構築した肝臓ではドナーの肝細胞と同程度かそれ以上の発現を示すことが確認されています。
製品
- Hu-liver TK-NOG マウス キメラ率70%以上
- Hu-liver TK-NOG マウス キメラ率40%以上
注)キメラ率はマウス血中のヒトアルブミン量から算出しています。
注)雄のみのご提供となります。 - ご購入にあたりまして使用同意書へのご署名が必要となります。
- またこのマウスモデルのご使用はカルタヘナ法の研究開発に係る遺伝子組み換え生物等にあたります。
弊社より情報提供書をお送りいたしますので、ご確認のうえ各機関のルールに従ってご使用ください。
研究用途
- 薬物動態解析モデル
- 薬物毒性試験モデル
- 安全性試験モデル
- ヒト疾患モデル(HBV/HCV等肝炎ウイルス感染モデル、肝臓がんモデル)
- 再生医療研究モデル
Hu-liver TK-NOGマウス表現型
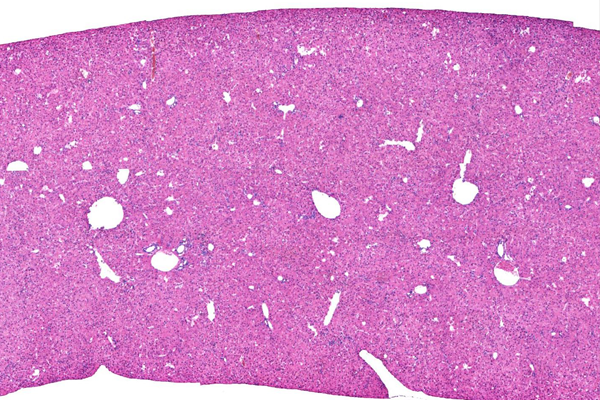
H&E 染色
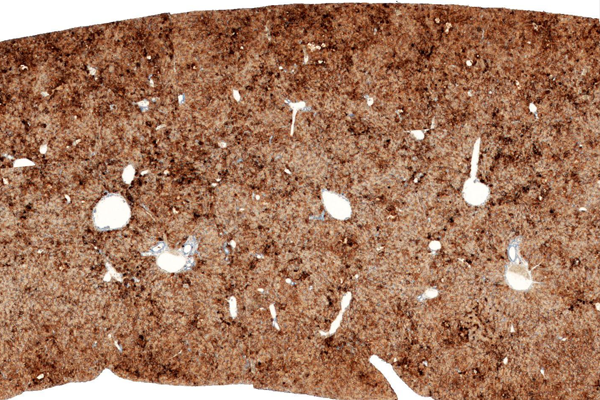
HLA 染色
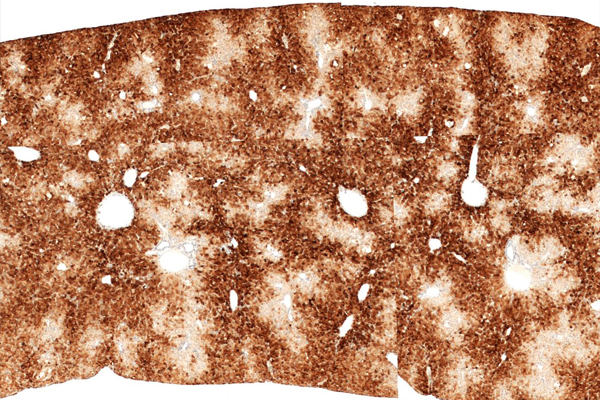
CYP3A4 染色
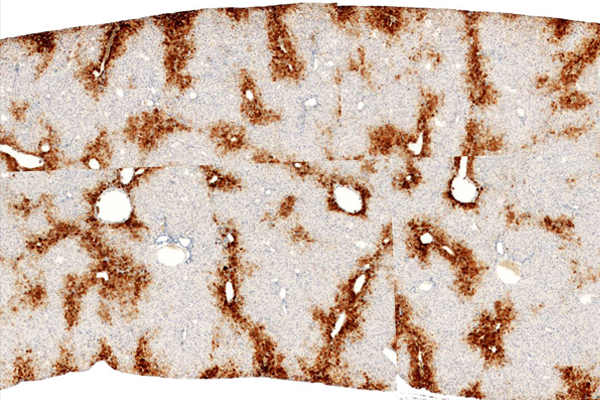
Glutamine Synthetase 染色
マウス内にヒト肝臓細胞が生着したキメラマウスを提供致します。
文献集
2024年度
A mechanistic biomarker investigation of fialuridine hepatotoxicity using the chimeric TK-NOG Hu-liver mouse model and in vitro micropatterned hepatocyte cocultures.(biomarker; fialuridine; humanized liver mouse; liver; mitochondrial DNA; p53.; Hu-Liver TK-NOG mouse)
Toxicol Res (Camb). 2024 Jan 11;13(1):tfad120. doi: 10.1093/toxres/tfad120. eCollection 2024 Feb.
Aslamkhan AG, Michna L, Podtelezhnikov A, Vlasakova K, Suemizu H, Ohnishi Y, Liu L, Lane P, Xu Q, Kuhls MC, Wang Z, Pacchione S, Erdos Z, Tracy RW, Koeplinger K, Muniappa N, Valentine J, Galijatovic-Idrizbegovic A, Glaab WE, Sistare FD, Lebron J.Hepatitis B virus-specific human stem cell memory T cells differentiate into cytotoxic T cells and eradicate HBV-infected hepatocytes in mice.(TK‐NOG mouse; cytotoxic T lymphocytes; hepatitis model; stem cell memory T cell (TSCM).)
FEBS Lett. 2024 Jun;598(11):1354-1365. doi: 10.1002/1873-3468.14842. Epub 2024 Apr 9.
Abe-Chayama H, Kawase T, Ichinohe T, Ishida Y, Tateno C, Hijikata M, Chayama K.Induction of drug metabolizing enzyme and drug transporter expression by antifungal triazole pesticides in human HepaSH hepatocytes.(Cytochromes P-450; Fungicides; HepaSH cells; Human hepatocytes; Transporters; Triazoles; Hu-liver TK-NOG)
Chemosphere. 2024 Oct:366:143474. doi: 10.1016/j.chemosphere.2024.143474. Epub 2024 Oct 5.
Anna Zerdoug , Marc Le Vée , Hélène Le Mentec, Jennifer Carteret , Elodie Jouan , Agnès Jamin , Béatrice Lopez , Shotaro Uehara , Yuichiro Higuchi , Nao Yoneda , Christophe Chesné , Hiroshi Suemizu , Olivier Fardel2023年度
The Unique Human N10-Glucuronidated Metabolite Formation from Olanzapine in Chimeric NOG-TKm30 Mice with Humanized Livers. (Olanzapine; NOG-TKm30)
Drug Metab Dispos . 2023 Apr;51(4):480 491. doi: 10.1124/dmd.122.001102. Epub 2023 Jan 9.
Uehara S, Higuchi Y, Yoneda N, Kato H, Yamazaki H, Suemizu H.Drug transporter expression and activity in cryopreserved human hepatocytes isolated from chimeric TK-NOG mice with humanized livers. (Cell culture; Cell suspension; Drug transporter; Human hepatocytes; Humanized liver mice; Transport activity; TK-NOG mice)
Toxicol In Vitro. 2023 Aug:90:105592. doi: 10.1016/j.tiv.2023.105592. Epub 2023 Apr 7.
Zerdoug A, Le Vée M, Uehara S, Jamin A, Higuchi Y, Yoneda N, Lopez B, Chesné C, Suemizu H, Fardel O.o-Toluidine metabolism and effects in the urinary bladder of humanized-liver mice.(Bladder; Humanized-liver mice; Liver; Metabolites; o-toluidine; NOG-TKm30)
Toxicology. 2023 Apr:488:153483. doi: 10.1016/j.tox.2023.153483. Epub 2023 Mar 3.
Yokota Y, Suzuki S, Gi M, Yanagiba Y, Yoneda N, Fujioka M, Kakehashi A, Koda S, Suemizu H, Wanibuchi H.HepaSH cells: Experimental human hepatocytes with lesser inter individual variation and more sustainable availability than primary human hepatocytes.(Chimeric mice; Cytochrome P450; Drugmetabolizing enzyme activity; Experimental human hepatocytes (EHH); HepaSH cells;Humanized liver mouse; Primary human hepatocytes (PHH))
Biochem Biophys Res Commun . 2023 Jun 30:663:132 141. doi: 10.1016/j.bbrc.2023.04.054. Epub 2023 Apr 25.
Shotaro Uehara Yuichiro Higuchi Nao Yoneda Ryoji Ito Takeshi Takahashi Norie Murayama Hiroshi Yamazaki Kazuhiro Murai Hayato Hikita Tetsuo Takehara Hiroshi SuemizuRoles of human cytochrome P450 3A4/5 in dexamethasone 6β-hydroxylation mediated by liver microsomes and humanized liver in chimeric mice metabolically suppressed with azamulin.(6β-hydroxydexamethasone ; Humanized liver mouse; Time dependent inhibitor)
Drug Metab Pharmacokinet 2023 Jun:50:100504. doi: 10.1016/j.dmpk.2023.100504. Epub 2023 Mar6.
Shotaro Uehara Makiko Shimizu Hiroshi Suemizu Hiroshi Yamazaki2022年度
The reconstituted 'humanized liver' in TK-NOG mice is mature and functional. (Hu-Liver TK-NOG)
Biochem Biophys Res Commun . 2011 Feb 18;405(3):405-10. doi: 10.1016/j.bbrc.2011.01.042. Epub 2011 Jan 14.
Masami Hasegawa , Kenji Kawai, Tetsuya Mitsui, Kenji Taniguchi, Makoto Monnai, Masatoshi Wakui, Mamoru Ito, Makoto Suematsu, Gary Peltz, Masato Nakamura, Hiroshi SuemizuHumanized liver TK-NOG mice with functional deletion of hepatic murine cytochrome P450s as a model for studying human drug metabolism.
Sci Rep. 2022 Sep 1;12(1):14907. doi: 10.1038/s41598-022-19242-0.
H, Ito M, Yamazaki H, Oshimura M, Kazuki Y, Suemizu H.Cytochrome P450-dependent drug oxidation activities and their expression levels in liver microsomes of chimeric TK-NOG mice with humanized livers.
Drug Metab Pharmacokinet. 2022 Jun;44:100454. doi: 10.1016/j.dmpk.2022.100454. Epub 2022 Feb 25.
Uehara S, Yoneda N, Higuchi Y, Yamazaki H, Suemizu H.An improved TK-NOG mouse as a novel platform for humanized liver that overcomes limitations in both male and female animals.
Drug Metab Pharmacokinet. 2022 Feb;42:100410. doi:10.1016/j.dmpk.2021.100410. Epub 2021 Jun 12.
Uehara S, Higuchi Y, Yoneda N, Kawai K, Yamamoto M, Kamimura H, Iida Y, Oshimura M, Kazuki Y, Yamazaki H, Hikita H, Takehara T, Suemizu H.Cytochrome P450s in chimeric mice with humanized liver.
Adv Pharmacol. 2022;95:307-328. doi: 10.1016/bs.apha.2022.05.004. Epub 2022 Jul 23.
Uehara S, Suemizu H, Yamazaki H.Expression and functional activity of cytochrome P450 enzymes in human hepatocytes with sustainable reproducibility for in vitro phenotyping studies. (Hu-Liver TK-NOG, human hepatocytes)
Adv Pharmacol. 2022;95:285-305. doi: 10.1016/bs.apha.2022.05.009. Epub 2022 Jun 30.
Bachour-El Azzi P, Chesné C, Uehara S.Formation of reactive metabolites of benzbromarone in humanized-liver mice.
Drug Metab Pharmacokinet. 2022 Aug 5;47:100467. doi: 10.1016/j.dmpk.2022.100467.
Cho N, Suemizu H, Kamimura H, Ohe T, Ito F, Akita H, Kobayashi K.2021年度
Novel hepatitis B virus infection mouse model using herpes simplex virus type 1 thymidine kinase transgenic mice. (HBV, )
J Gastroenterol Hepatol. 2021 Mar;36(3):782-789. doi: 10.1111/jgh.15142. Epub 2020 Jul 7.
Kanbe A, Ishikawa T, Hara A, Suemizu H, Nanizawa E, Tamaki Y, Ito H.Methyl-hydroxylation and subsequent oxidation to produce carboxylic acid is the major metabolic pathway of tolbutamide in chimeric TK-NOG mice transplanted with human hepatocytes.
Xenobiotica. 2021 May;51(5):582-589. doi: 10.1080/00498254.2021.1875515. Epub 2021 Feb 18.
Uehara S, Yoneda N, Higuchi Y, Yamazaki H, Suemizu H.Characterization of plasma protein binding in two mouse models of humanized liver, PXB mouse and humanized TK-NOG mouse.
Xenobiotica. 2021 Jan;51(1):51-60. doi:10.1080/00498254.2020.1808735. Epub 2020 Aug 25.
ki Miyamoto, Yohei Kosugi, Shinji Iwasaki, Ikumi Chisaki, Sayaka Nakagawa, Nobuyuki Amano, Hideki HirabayashiIn Vivo Functional Analysis of Nonconserved Human lncRNAs Using a Humanized Mouse Model. (TK-NOG, Nonconserved Human lncRNAs )
Methods Mol Biol. 2021;2254:339-347. doi: 10.1007/978-1-0716-1158-6_21.
Ma Y, Jiang CF, Li P, Cao H.Plasmodium falciparum-infected humanized mice: a viable preclinical tool.
Immunotherapy. 2021 Nov;13(16):1345-1353. doi: 10.2217/imt-2021-0102. Epub 2021 Aug 23.
Tyagi RK.2020年度
Human Aldehyde Oxidase 1-Mediated Carbazeran Oxidation in Chimeric TK-NOG Mice Transplanted with Human Hepatocytes.
Drug Metab Dispos. 2020 Jul;48(7):580-586. doi: 10.1124/dmd.120.091090. Epub 2020 May 1.
Uehara S, Yoneda N, Higuchi Y, Yamazaki H, Suemizu H.Metabolism of desloratadine by chimeric TK-NOG mice transplanted with human hepatocytes.
Xenobiotica . 2020 Jun;50(6):733-740. doi: 10.1080/00498254.2019.1688892. Epub 2019 Nov 12.
Uehara S, Yoneda N, Higuchi Y, Yamazaki H, Suemizu H.A long-acting 3TC ProTide nanoformulation suppresses HBV replication in humanized mice. (suppresses HBV replication)
Nanomedicine. 2020 Aug;28:102185. doi: 10.1016/j.nano.2020.102185. Epub 2020 Mar 24.
Wang W, Smith N, Makarov E, Sun Y, Gebhart CL, Ganesan M, Osna NA, Gendelman HE, Edagwa BJ, Poluektova LY.Attenuated P. falciparum Parasite Shows Cytokine Variations in Humanized Mice.(P. falciparum , TK-NOG, (hRBC-TK-NOG, P. falciparum )
Front Immunol. 2020 Sep 11;11:1801. doi: 10.3389/fimmu.2020.01801. eCollection 2020.
Zhang LL, Li JL, Ji MX, Tian D, Wang LY, Chen C, Tian M.2019年度
Predictability of human pharmacokinetics of diisononyl phthalate (DINP) using chimeric mice with humanized liver. (PK/DINP, TK-NOG)
Xenobiotica. 2019 Nov;49(11):1311-1322. doi: 10.1080/00498254.2018.1564087. Epub 2019 Jun 21.
Iwata H, Goto M, Sakai N, Suemizu H, Yamazaki H.Establishment of the Dual Humanized TK-NOG Mouse Model for HIV-associated Liver Pathogenesis.
J Vis Exp. 2019 Sep 11;(151):10.3791/58645. doi: 10.3791/58645.
Dagur RS, Wang W, Makarov E, Sun Y, Poluektova LY.Combinations of two drugs among NS3/4A inhibitors, NS5B inhibitors and non-selective antiviral agents are effective for hepatitis C virus with NS5A-P32 deletion in humanized-liver mice. (DAA; Humanized liver mice; Recombinant HCV; RASs; Hu-Livr TK-NOG).
J Gastroenterol. 2019 May;54(5):449-458. doi: 10.1007/s00535-018-01541-x. Epub 2019 Jan 25.
Doi A, Hikita H, Kai Y, Tahata Y, Saito Y, Nakabori T, Yamada R, Kodama T, Sakamori R, Murayama A, Nitta S, Asahina Y, Suemizu H, Tatsumi T, Kato T, Takehara T.2018年度
Humanized Mice Are Instrumental to the Study of Plasmodium falciparum Infection. (TK-NOG, Plasmodium falciparum Infection)
Front Immunol. 2018 Dec 13;9:2550. doi: 10.3389/fimmu.2018.02550. eCollection 2018.
Tyagi RK, Tandel N, Deshpande R, Engelman RW, Patel SD, Tyagi P.Human plasma metabolic profiles of benzydamine, a flavin-containing monooxygenase probe substrate, simulated with pharmacokinetic data from control and humanized-liver mice.(TK-NOG, metabolic profiles of benzydamine)
Xenobiotica. 2018 Feb;48(2):117-123. doi:10.1080/00498254.2017.1288280. Epub 2017 Feb 21.
Yamazaki-Nishioka M, Shimizu M, Suemizu H, Nishiwaki M, Mitsui M, Yamazaki H.Human hepatocyte depletion in the presence of HIV-1 infection in dual reconstituted humanized mice. (humanized mice, HSC-TK-NOG, HIV)
Biol Open. 2018 Feb 13;7(2):bio029785. doi: 10.1242/bio.029785.
Dagur RS, Wang W, Cheng Y, Makarov E, Ganesan M, Suemizu H, Gebhart CL, Gorantla S, Osna N, Poluektova LY.2017年度
Human fetal liver cultures support multiple cell lineages that can engraft immunodeficient mice.(TK-NOG, fetal liver)
Open Biol. 2017 Dec;7(12):170108. doi: 10.1098/rsob.170108.
Fomin ME, Beyer AI, Muench MO.Observation of Clinically Relevant Drug Interaction in Chimeric Mice with Humanized Livers: The Case of Valproic Acid and Carbapenem Antibiotics. (Hu-Liver TK-NOG, DDI)
Eur J Drug Metab Pharmacokinet. 2017 Dec;42(6):965-972. doi: 10.1007/s13318-017-0413-2.
Suzuki E, Koyama K, Nakai D, Goda R, Kuga H, Chiba K.Metabolic profiles of pomalidomide in human plasma simulated with pharmacokinetic data in control and humanized-liver mice.(PBPK modeling, Hu-Liver TK-NOG).
Xenobiotica. 2017 Oct;47(10):844-848. doi: 10.1080/00498254.2016.1247218. Epub 2016 Nov 16.
Shimizu M, Suemizu H, Mitsui M, Shibata N, Guengerich FP, Yamazaki H.Simulation of human plasma concentration-time profiles of the partial glucokinase activator PF-04937319 and its disproportionate N-demethylated metabolite using humanized chimeric mice and semi-physiological pharmacokinetic modeling. (Chimeric mouse; MIST guidance; cytochrome P450; disproportionate metabolite; humanized liver; semi-physiological pharmacokinetics. TK-NOG)
Xenobiotica. 2017 May;47(5):382-393. doi:10.1080/00498254.2016.1199063. Epub 2016 Jul 7.
Kamimura H, Ito S, Chijiwa H, Okuzono T, Ishiguro T, Yamamoto Y, Nishinoaki S, Ninomiya SI, Mitsui M, Kalgutkar AS, Yamazaki H, Suemizu H.Demethylase JMJD6 as a New Regulator of Interferon Signaling: Effects of HCV and Ethanol Metabolism.(JMJD6, HCV/Ethanol Metabolism, Hu-Liver TK-NOG)
Cell Mol Gastroenterol Hepatol. 2017 Oct 16;5(2):101-112. doi: 10.1016/j.jcmgh.2017.10.004. eCollection 2018.
Ganesan M, Tikhanovich I, Vangimalla SS, Dagur RS, Wang W, Poluektova LI, Sun Y, Mercer DF, Tuma D, Weinman SA, Kharbanda KK, Osna NA.2016年度
Functional polymer-dependent 3D culture accelerates the differentiation of HepaRG cells into mature hepatocytes. (Hepa-RG, TK-NOG)
Hepatol Res. 2016 Sep;46(10):1045-57. doi: 10.1111/hepr.12644. Epub 2016 Feb 7.
Higuchi Y, Kawai K, Kanaki T, Yamazaki H, Chesné C, Guguen-Guillouzo C, Suemizu H.Sodium taurocholate cotransporting polypeptide inhibition efficiently blocks hepatitis B virus spread in mice with a humanized liver. (TK-NOG, HBV)
Sci Rep. 2016 Jun 9;6:27782. doi: 10.1038/srep27782.
Nakabori T, Hikita H, Murai K, Nozaki Y, Kai Y, Makino Y, Saito Y, Tanaka S, Wada H, Eguchi H, Takahashi T, Suemizu H, Sakamori R, Hiramatsu N, Tatsumi T, Takehara T.Human plasma concentrations of cytochrome P450 probe cocktails extrapolated from pharmacokinetics in mice transplanted with human hepatocytes and from pharmacokinetics in common marmosets using physiologically based pharmacokinetic modeling. (Hepatic clearance; P450 substrates; PBPK modeling, HY-Liver TK-NOG),
Xenobiotica. 2016 Dec;46(12):1049-1055. doi: 10.3109/00498254.2016.1147102. Epub 2016 Feb 25.
Utoh M, Suemizu H, Mitsui M, Kawano M, Toda A, Uehara S, Uno Y, Shimizu M, Sasaki E, Yamazaki H.2015年度
Humanized thymidine kinase-NOG mice can be used to identify drugs that cause animal-specific hepatotoxicity: a case study with furosemide.
J Pharmacol Exp Ther . 2015 Jul;354(1):73-8. doi: 10.1124/jpet.115.224493. Epub 2015 May 11.
Xu D, Michie SA, Zheng M, Takeda S, Wu M, Peltz G.Human Cytotoxic T Lymphocyte-Mediated Acute Liver Failure and Rescue by Immunoglobulin in Human Hepatocyte Transplant TK-NOG Mice.
J Virol. 2015 Oct;89(19):10087-96. doi: 10.1128/JVI.01126-15. Epub 2015 Aug 5.
Uchida T, Hiraga N, Imamura M, Tsuge M, Abe H, Hayes CN, Aikata H, Ishida Y, Tateno C, Yoshizato K, Ohdan H, Murakami K, Chayama K.Chimeric TK-NOG mice: a predictive model for cholestatic human liver toxicity.
J Pharmacol Exp Ther. 2015 Feb;352(2):274-80. doi: 10.1124/jpet.114.220798. Epub 2014 Nov 25.
Takeda ST, Guan Y, Guo Y, Peltz G.Formation of the accumulative human metabolite and human-specific glutathione conjugate of diclofenac in TK-NOG chimeric mice with humanized livers.
Drug Metab Dispos. 2015 Mar;43(3):309-16. doi: 10.1124/dmd.114.061689. Epub 2014 Dec 11.
Kamimura H, Ito S, Nozawa K, Nakamura S, Chijiwa H, Nagatsuka S, Kuronuma M, Ohnishi Y, Suemizu H, Ninomiya S.Plasmodium falciparum full life cycle and Plasmodium ovale liver stages in humanized mice. (TK-NOG, Plasmodium falciparum full life cycle ),
Nat Commun. 2015 Jul 24;6:7690. doi: 10.1038/ncomms8690.
Soulard V, Bosson-Vanga H, Lorthiois A, Roucher C, Franetich JF, Zanghi G, Bordessoulles M, Tefit M, Thellier M, Morosan S, Le Naour G, Capron F, Suemizu H, Snounou G, Moreno-Sabater A, Mazier D.Emergence of hepatitis C virus NS5A L31V plus Y93H variant upon treatment failure of daclatasvir and asunaprevir is relatively resistant to ledipasvir and NS5B polymerase nucleotide inhibitor GS-558093 in human hepatocyte chimeric mice. (HCV infection, HU-Liver TK-NOG, (Asunaprevir/daclatasvir; Ledipasvir/sofosbuvir; Resistance-associated variants (RAVs)).
J Gastroenterol. 2015 Nov;50(11):1145-51. doi: 10.1007/s00535-015-1108-6. Epub 2015 Jul 25.
Kai Y, Hikita H, Tatsumi T, Nakabori T, Saito Y, Morishita N, Tanaka S, Nawa T, Oze T, Sakamori R, Yakushijin T, Hiramatsu N, Suemizu H, Takehara T.